Introduction
Calorimetry is a method of measuring energy changes in chemical reactions, and is based on two fundamental laws:
To carry out calorimetry, we must assume the following things:
- The First Law of Thermodynamics (The Law of Conservation of Energy): the energy of an isolated system is constant; the energy can be transformed into different forms, but cannot be created or destroyed.
- Hess’s Law of Constant Heat Summation: regardless of the multiple intermediate stages or steps of a reaction, the total enthalpy change of the reaction is the sum of all changes.
To carry out calorimetry, we must assume the following things:
- No heat is transferred to or from the surroundings (reaction is a closed system)
- No heat is absorbed or released from the equipment used to carry out the experiment
- The density and heat capacity of any aqueous solution is equal to that of pure water (d = 1.00 g/mL and c = 4.18 J/g * °C)
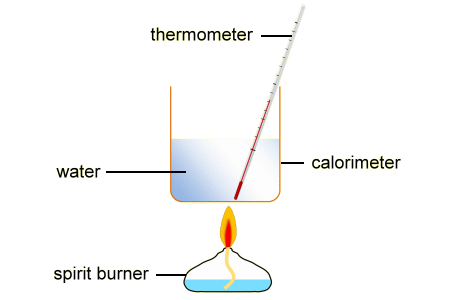
Why do we need a calorimeter?
Calorimeter is an apparatus that is generally used to measure the amount of heat energy given off in a chemical reaction. It prevents the heat from escaping and keeps it within the system to get an accurate measurement. Whereas thermometer within the calorimeter will measure the temperature change of the reaction.
Calculating Heat using Calorimetry
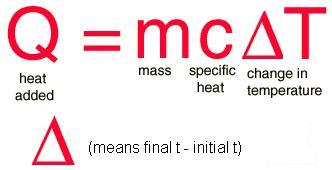
Q = Amount of heat energy (Joules = J)
m = mass (grams = g)
c = specific heat capacity (J/g⋅oC)
ΔT = change in temperature (Final temperature - Initial temperature)
m = mass (grams = g)
c = specific heat capacity (J/g⋅oC)
ΔT = change in temperature (Final temperature - Initial temperature)