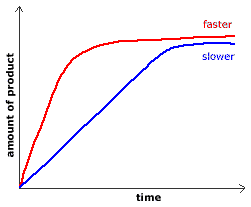
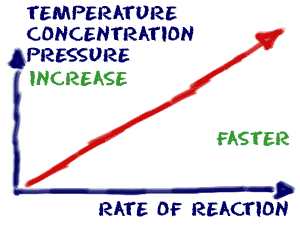
Introduction to Rates of Reaction and Rate Laws
The following lesson involves some heavy concepts! We recommend that students use this resource in groups of 2 or 3, in order for collaboration and discussion. Numerous resources have been adapted to make this lesson a perfect fit for a 12U class.
The rate of a reaction can be measured by the rate at which a reactant is used up, or the rate at which a product is formed. The temperature, concentration, pressure of reacting gases, surface area of reacting solids, and the use of catalysts, are all factors which affect the rate of a reaction.
Collision Theory
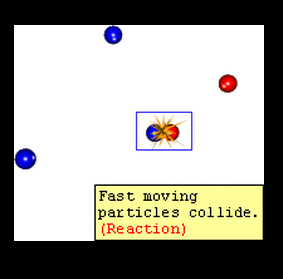
There is another big idea for rates of reaction called collision theory. The collision theory says that as more collisions in a system occur, there will be more combinations of molecules bouncing into each other. If you have more possible combinations there is a higher chance that the molecules will complete the reaction. The reaction will happen faster which means the rate of that reaction will increase.
How Can We Increase Collisions?
This is a great point to stop and ask the class some ways in which they think they could increase a rate of reaction! They've just learned the basics of collision theory, and now can attempt to apply their knowledge. This ties directly into the specific expectations.
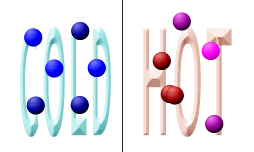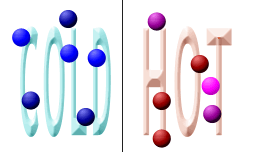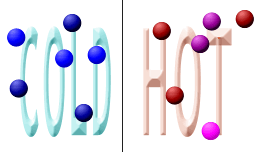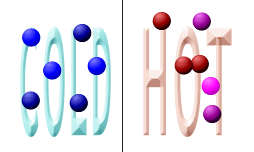
Temperature: When you raise the temperature of a system, the molecules bounce around a lot more. They have more energy. When they bounce around more, they are more likely to collide. That fact means they are also more likely to combine. When you lower the temperature, the molecules are slower and collide less. That temperature drop lowers the rate of the reaction. To the chemistry lab! Sometimes you will mix solutions in ice so that the temperature of the system stays cold and the rate of reaction is slower.
Concentration: If there is more of a substance in a system, there is a greater chance that molecules will collide and speed up the rate of the reaction. If there is less of something, there will be fewer collisions and the reaction will probably happen at a slower speed. Sometimes, when you are in a chemistry lab, you will add one solution to another. When you want the rate of reaction to be slower, you will add only a few drops at a time instead of the entire beaker.
Pressure: Pressure: Pressure affects the rate of reaction, especially when you look at gases. When you increase the pressure, the molecules have less space in which they can move. That greater density of molecules increases the number of collisions. When you decrease the pressure, molecules don't hit each other as often and the rate of reaction decreases.
Concentration: If there is more of a substance in a system, there is a greater chance that molecules will collide and speed up the rate of the reaction. If there is less of something, there will be fewer collisions and the reaction will probably happen at a slower speed. Sometimes, when you are in a chemistry lab, you will add one solution to another. When you want the rate of reaction to be slower, you will add only a few drops at a time instead of the entire beaker.
Pressure: Pressure: Pressure affects the rate of reaction, especially when you look at gases. When you increase the pressure, the molecules have less space in which they can move. That greater density of molecules increases the number of collisions. When you decrease the pressure, molecules don't hit each other as often and the rate of reaction decreases.
A Quick, Corny Game
This game loosely displays the idea that heat can have an effect on a reaction! This ties into applications of collision theory.
Rate Laws
This is possibly the most important concept for the rest of the unit and one that students may have trouble with! We recommend going through some of the examples provided in detail with the class, then working on them together, and finally allowing them to work on their own. This is a basic I Do-We Do-You Do structure.
Chemical Kinetics is the study of ways to make a reaction go towards the products faster or slower. The pace at which the reaction occurs is termed the reaction rate. The reaction rate is the change in amount of reactants (or products) over time.
Generally, we show these reaction rates with the following units:
Reaction Rate = [Concentration of a particular substance] / [Unit of time, generally seconds]
The equation for this would be:
Rate of Reaction = The Change in Concentration / The Change in Time
A reaction rate will always be shown as positive. Why might this be? This is a great time to stop the class and ask for reasons to get them to think about the system.
Chemical Kinetics is the study of ways to make a reaction go towards the products faster or slower. The pace at which the reaction occurs is termed the reaction rate. The reaction rate is the change in amount of reactants (or products) over time.
Generally, we show these reaction rates with the following units:
Reaction Rate = [Concentration of a particular substance] / [Unit of time, generally seconds]
The equation for this would be:
Rate of Reaction = The Change in Concentration / The Change in Time
A reaction rate will always be shown as positive. Why might this be? This is a great time to stop the class and ask for reasons to get them to think about the system.
Example: What is the overall rate of production of nitrogen dioxide in the system shown below if the concentration of nitrogen dioxide changes from 0.24mol/L to 0.78mol/L in 5 minutes?
N2(g) + 2O₂(g) -> 2NO₂(g)
N2(g) + 2O₂(g) -> 2NO₂(g)
The following resources are for differentiating between INSTANTANEOUS and AVERAGE rates of change
|
|
|
Resources
-http://www.businessinsider.com/14-awesome-gifs-that-will-make-you-wish-you-paid-attention-in-chemistry-2013-5
-http://www.chem4kids.com/files/react_rates.html
-http://www.bbc.co.uk/schools/gcsebitesize/science/add_edexcel/chemical_reactions/ratesrev1.shtml
-http://www.ddsb.ca/school/dce/Programs/lah/lahdownload/sch4u/Documents/SCH4U%20-%20Unit%203%20-%20Version%20C.pdf
-http://www.chem4kids.com/files/react_rates.html
-http://www.bbc.co.uk/schools/gcsebitesize/science/add_edexcel/chemical_reactions/ratesrev1.shtml
-http://www.ddsb.ca/school/dce/Programs/lah/lahdownload/sch4u/Documents/SCH4U%20-%20Unit%203%20-%20Version%20C.pdf