Introduction to Modelling Rates
The lesson will begin with an introduction to Potential Energy Diagrams, which will be a theme throughout. This is a necessary component of understanding Rates of Reaction and is important information for the Project in the next lesson!
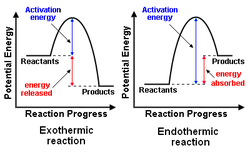
Potential Energy Diagrams: A potential energy diagram is a graphical representation of the energy transferred during a physical or chemical change.
Activation Energy: The minimum energy required for effective collisions to occur.
Activated Complex(es): An unstable arrangement of atoms that represents the maximum potential energy.
Activation Energy: The minimum energy required for effective collisions to occur.
Activated Complex(es): An unstable arrangement of atoms that represents the maximum potential energy.
Temperature is a measure of the kinetic energy of a substance. So if you heat a reaction you are giving it more kinetic energy so they can have more effective collisions. Check out the above video! Notice how the boiled of the water is made faster by applying it to hot sand! A super cool trick.
Looking at the Factors Affecting Rates of Reaction
In this section, we build a knowledge base and an understanding of how potential energy diagrams change based on a the different factors affecting rates. For each category, stop and discuss with the class that potential energy diagrams may look like for a high concentration solution compared to a low concentration solution. Also high surface area vs low surfaces area. For catalysts, and example is provided.
Explaining Concentration & Surface Area
Concentration – if there are more particles around then they are more likely to undergo effective collisions. Surface Area – if there is more space for the particles to interact they are more likely to undergo effective collision.
Concentration – if there are more particles around then they are more likely to undergo effective collisions. Surface Area – if there is more space for the particles to interact they are more likely to undergo effective collision.

Explaining Catalysts
Catalysts – reduce the activation energy…so it takes less energy for the reaction to happen.
Catalysts – reduce the activation energy…so it takes less energy for the reaction to happen.

Reaction Mechanisms
Reactions don’t happen in one step! Introduce students to the concept of multi-step reactions. Bring back the Iodine Clock reaction as an example of a multi-step reaction.
Elementary Step – a step involving one, two or three entity collisions
Reaction Mechanism – a series of elementary steps that give the overall equation
Reactions don’t happen in one step! Introduce students to the concept of multi-step reactions. Bring back the Iodine Clock reaction as an example of a multi-step reaction.
Elementary Step – a step involving one, two or three entity collisions
Reaction Mechanism – a series of elementary steps that give the overall equation
A couple more important terms:
Rate determining step – the slowest step in a reaction mechanism
Reaction intermediates – a substance produced in one elementary step and used up in another reaction (i.e. not in the final equation)
Reaction intermediates – a substance produced in one elementary step and used up in another reaction (i.e. not in the final equation)
Resources:
-http://en.citizendium.org/images/f/fb/ExoEndo_Reax.png
-http://mdriscoll.pbworks.com/w/file/fetch/65067720/6.3%20%26%206.6%20%20Explaining%20Reaction%20Rate%20%26%20Reaction%20Mechanisms.pdf
-http://www.proprofs.com/quiz-school/user_upload/ckeditor/6_3a.png
-http://www.softschools.com/notes/ap_chemistry/images/multistep_reactions_img_1.png
-http://en.citizendium.org/images/f/fb/ExoEndo_Reax.png
-http://mdriscoll.pbworks.com/w/file/fetch/65067720/6.3%20%26%206.6%20%20Explaining%20Reaction%20Rate%20%26%20Reaction%20Mechanisms.pdf
-http://www.proprofs.com/quiz-school/user_upload/ckeditor/6_3a.png
-http://www.softschools.com/notes/ap_chemistry/images/multistep_reactions_img_1.png
