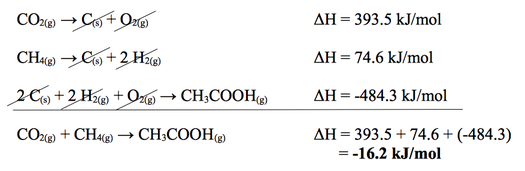What is Hess' Law?
The value of ΔH in any given reaction can be written in multiple steps is the sum of ΔH of the individual steps.
Hess' Law: Method 1
- Identify and balance the chemical equation
- Identify individual steps of the equation
- Reverse any steps of the equation so that the position of the reactants and products add up to the overall equation
- Multiply the step equation by the appropriate coefficient to match the original equation (remember to also multiply ΔH by the same coefficient)
- Add up the step equations and their ΔH values. The sum of the modified intermediate step equations should be identical to the overall equation.
Hess' Law: Method 2
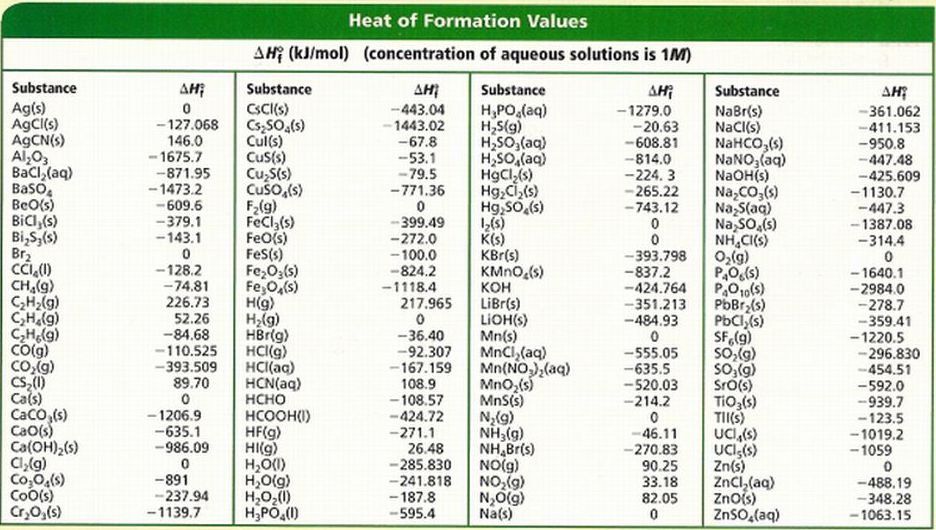
Identify the ΔH of formation for each reactant and product and solve using the following equation:
Example Question
In the question above, we see that carbon dioxide gas is reacting wiht methane gas to create acetic acid. We know that carbon dioxide and methane are reactants and using the enthalpy of formation chart (found below), we can find their average ΔH of formation values. *Remember to change the ΔH value since they are on the reactant side rather than the product side (change the negative value to positive or vice versa). Acetic acid is the product of this chemical reaction therefore ΔH value remains unchanged. Add the ΔH values together to get ΔH value for your overall equation.