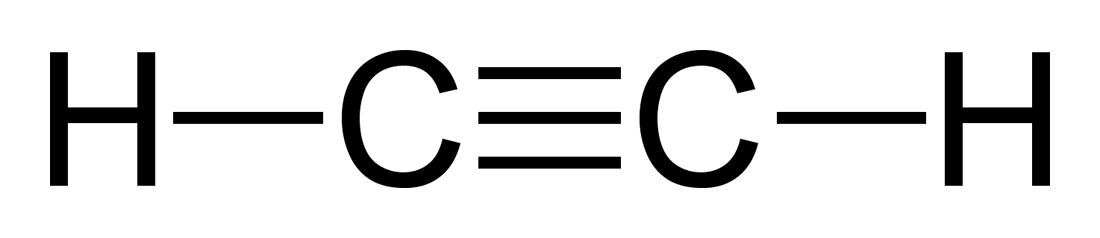Naming Basic Hydrocarbons & an Introduction to Organic Chemistry
Organic is a word that often gets thrown around in the media, on food labels, etc.
What does the word "organic" mean to you?
What does the word "organic" mean to you?
- Give your students about 2-3 minutes to brainstorm together in whatever way they like (mind map, list, basic discussion), using Think, Pair, Share
- A cool way to get students to display their thoughts is by using Mentimeter to instantly make a word cloud of their thoughts about the meaning of the word "organic".
This here, for example, is a word cloud generated by adding in the Wikipedia article on organic food. This gives you an idea of what your students might put down.
|
Next, explain to them that organic probably doesn't mean what they thought based on media perceptions and food blogs.
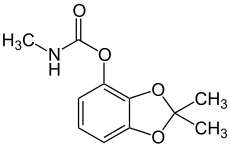
The definition of organic with respect to chemistry is: a compound that is based on carbon. That's it. A bit underwhelming, really. This means that the vast majority of chemical compounds on earth are considered to be organic. Take, for example, bendiocarb: |
Bendiocarb is an insecticide, and is also considered to be an organic compound, due to the large amount of carbon it contains (every stick junction where there is no other specified atom such as oxygen or nitrogen is a carbon).
Previously, organic compounds were only those that were from living things, but in 1828, a German chemist by the name of Friedrich Wohler successfully got urea (a compound found in urine), from ammonium cyanate (a non-living susbtance). Since an inorganic compound gave way to an organic one, the definition was thus changed to any compound with a heavy emphasis on carbon. Cool, huh?
Previously, organic compounds were only those that were from living things, but in 1828, a German chemist by the name of Friedrich Wohler successfully got urea (a compound found in urine), from ammonium cyanate (a non-living susbtance). Since an inorganic compound gave way to an organic one, the definition was thus changed to any compound with a heavy emphasis on carbon. Cool, huh?
A great tie-in into the Structure & Properties of Matter unit is having a discussion with your students as to why carbon is in so many compounds. Consider its electron configuration: how many bonds can carbon form? Four. This means that if it is saturated and forms only single bonds, it will make the tetrahedral VSEPR shape. Because of this unique property of carbon, more organic compounds exist on earth than inorganic compounds.
What kinds of bonds can carbon make?
- Students should say that it can form single, double and triple bonds. Carbon is a very versatile atom.
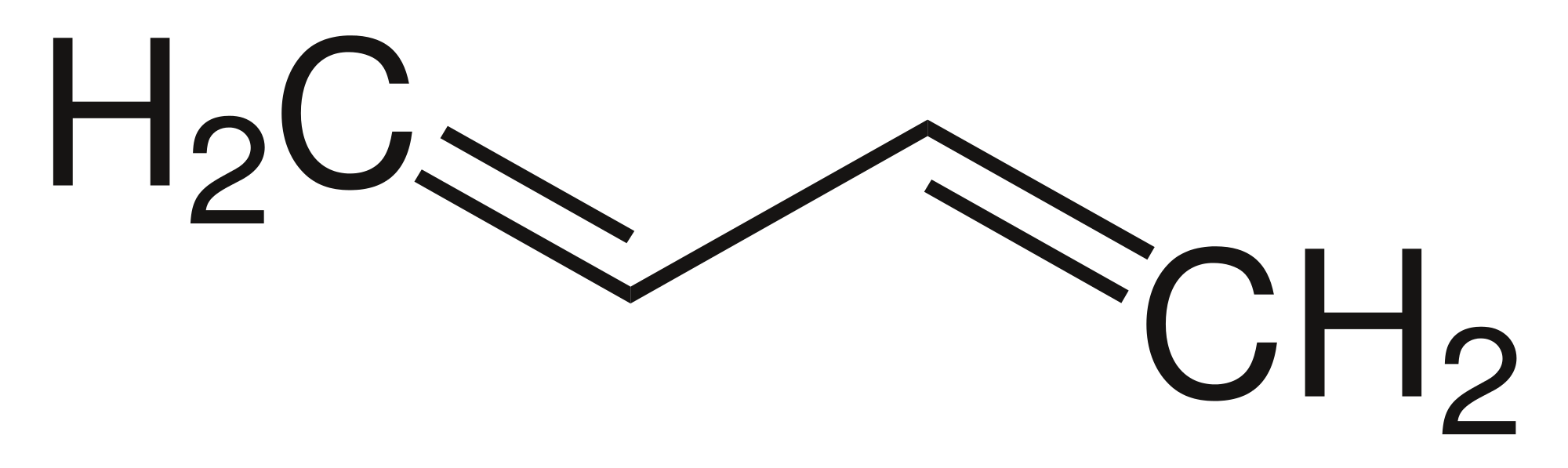
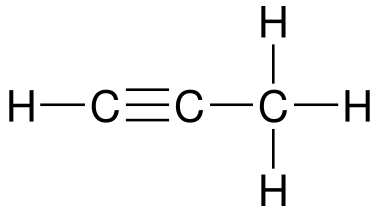
Consider 3 organic compounds:
What are some similarities between these three compounds? What about some differences? Some possible answers you may get from your students:
- All three have two carbon atoms
- All three have a differing number of hydrogen atoms
- The carbon atoms are either single, double, or triple bonded together
The one on the left (ethane) is what we call saturated. All of the carbon atoms have the maximum number of bonds (four), there are only single bonds.
- A good way to teach your students about saturated compounds is that "saturated" and "single" both start with "S".
Where have you heard the terms "saturated" and "unsaturated" before?
- Your students should have heard of saturated and unsaturated fats
- Saturated fats are straight chains, while unsaturated fats have "kinks" in them
- We will come back to this later on when we discuss alkenes
Basic Naming
Why is it useful to have a specific naming convention? Universality!
- When a chemist in Brazil discusses methanoic acid and its potential uses with a chemist in Croatia, although they don't speak the same language, they can be assured they are discussing the same chemical
- This has implications in safety (i.e., if research is released about the new health benefits of propanol, it is important that someone doesn't mistake this for methanol and possibly make many people sick)
Some compounds have more than one acceptable name: an IUPAC name and a common name
- e.g., formic acid and methanoic acid are the same
When naming compounds, it is important to remember that certain things take precedence over others. In general, follow this pattern:
prefix + root + suffix
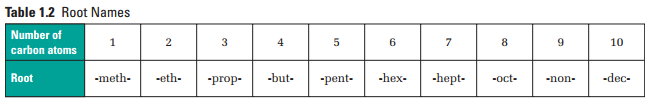
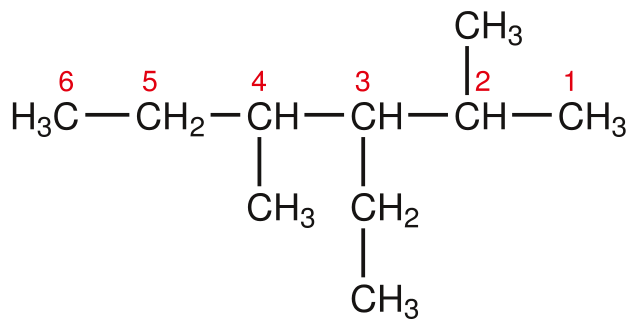
To name the root, you first need to know the length of the main chain, or the number of carbon atoms.
When determining the name:
When determining the name:
- Always count the longest chain (you will see what I mean in a minute)
- Always include any multiple bonds
- The functional group has to have the lowest possible number
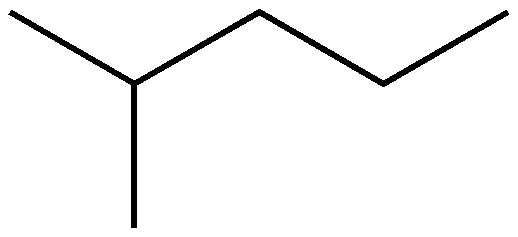
Here are the steps needed to take to name hydrocarbons, according to the McGraw-Hill Ryerson Chemistry 12 textbook, along with the root names dependent on the number of carbons in the main chain:
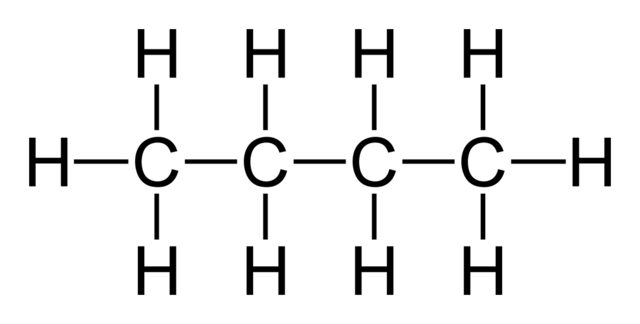
Alkanes
The natural starting point for introducing hydrocarbons is the most simple: the alkane.
What is a hydrocarbon? Have your students think about the word and decipher its meaning.
What is a hydrocarbon? Have your students think about the word and decipher its meaning.
- A compound that contains carbon and hydrogen
- Only single bonds, so it is saturated
- The suffix of an alkane is always -ane, and the root name is determined by the chain length, or the number of carbons
- They can also be cyclic
Let's try to name some examples together:
Alkenes
Given the name of an alkene compared to an alkane, what do you think an alkene is?
- Hydrocarbon which contains at least one double bond
- Always include the double bond in the main chain when naming alkenes
- The suffix is -ene
- These can also be cyclic
Cis means both the carbons are on the same side of the double bond, while trans means they are on opposite sides.
Unsaturated fats can be cis or trans
Unsaturated fats can be cis or trans
- Trans fats essentially make straight chain alkenes, thus are dangerous like saturated fats
- There are no naturally-occurring trans fats, but saturated fats exist in nature (i.e., coconuts, avocados, etc.)
Notice that in these two examples, we have to denote the position of the double bonds with a number. Also, we have to mention in the name that there is more than one double bound by putting "di-" in the middle of "butene".
Alkynes
If alkanes have only single bonds and alkenes have double bonds, what do alkynes have?
- Triple bonds (at least one)
- Use the -yne ending in naming
- Your students should say no
- Why not?
- The carbons would have more than 4 bonds
- The carbons would have more than 4 bonds
Have students build the compounds you discuss using molecular model kits, so they can get an idea of the confirmation of the organic compounds and have a better understanding of how they look in 3D, rather than only in 2D on a piece of paper. With this more hands-on approach, students will be able to better visualize the structures later on, even when they don't have the model kits in front of them. This is a good strategy to use throughout the whole unit. Allow students to do this on tests so they are better prepared for post-secondary education, as well.
Here are some videos you can show in your class to your students to help them out. Crash Course Chemistry has 46 videos about all sorts of chemistry topics with great visuals and are a good summary/consolidation to any lesson.
|
|
|
Homework:
Chemistry 12, pg 16 & 17, Q 5-9
Chemistry 12, pg 16 & 17, Q 5-9